Uses of Hyperbaric Oxygen Therapy in Serious Infections: Gas Gangrene, NSTI, and Osteomyelitis
Three of the most dangerous infections share one biological vulnerability in common: they thrive in hypoxic tissue. The uses of hyperbaric oxygen therapy in infection are built entirely around that vulnerability.
[IMAGE: uses-of-hyperbaric-oxygen-therapy-infection-gas-gangrene-nsti-osteomyelitis.jpg | Alt: uses of hyperbaric oxygen therapy — infection gas gangrene NSTI osteomyelitis HBOT anaerobic]
These infection uses are not widely known outside specialist infectious disease and emergency medicine circles.
They should be. Three of the most lethal infections that present to Indian hospitals — gas gangrene, necrotising soft tissue infections, and refractory osteomyelitis — have formal FDA and UHMS recognition as HBOT indications. Each shares a core biological mechanism: the infection thrives in hypoxic tissue, and HBOT destroys that environment.
This article covers all three infection uses in one place — the mechanism, the evidence, and the clinical context that determines when HBOT belongs in the treatment plan alongside surgery and antibiotics.
For the foundational mechanism of how HBOT oxygenates hypoxic tissue, visit How HBOT Works. For the full 14-indication overview, see our HBOT uses guide.
Why infection is an HBOT indication: The three infection uses of HBOT share one mechanism — the pathogens responsible (Clostridia, mixed anaerobes, Staphylococcus in biofilm) all have reduced virulence or direct lethality in high-oxygen tissue environments. HBOT raises tissue oxygen to levels that kill obligate anaerobes, suppress toxin production, restore leukocyte bactericidal function, and potentiate antibiotic efficacy. None of these effects are achievable at normal atmospheric pressure.
Infection Use 1 — Uses of Hyperbaric Oxygen Therapy in Gas Gangrene
Gas gangrene — clostridial myonecrosis — is caused by Clostridium perfringens and related species. These are obligate anaerobes: they die in oxygen. The problem is that they create the hypoxic environment they need to survive by destroying the microvascular supply of the tissue they infect.
The infection spreads at several centimetres per hour. Alpha-toxin production destroys tissue independently of bacterial count. And the hypoxic environment disables leukocyte killing — the immune mechanism that would otherwise contain the spread.
HBOT at 2.5 to 3.0 ATA raises tissue oxygen to directly bactericidal levels, suppresses alpha-toxin production by approximately 50%, and restores leukocyte function. Combined with surgery and antibiotics, it reduces mortality from 20–30% to single figures. The detailed three-mechanism evidence is covered in our article on HBOT treatment for gas gangrene.
| Gas Gangrene Evidence | Surgery alone: 20–30% mortality. Surgery plus antibiotics plus HBOT at 2.5–3.0 ATA: 5–10% mortality in centres with rapid access. Three sessions in the first 24 hours is standard — the intensive first-day protocol is what makes the difference. Every hour from diagnosis to first HBOT session allows further bacterial spread and toxin accumulation. |
Infection Use 2 — Uses of Hyperbaric Oxygen Therapy in Necrotising Fasciitis
Necrotising soft tissue infections — including necrotising fasciitis, Fournier’s gangrene, and other variants — are typically caused by mixed aerobic and anaerobic bacteria acting synergistically. The anaerobic component creates the hypoxic tissue environment. The aerobic component produces toxins that further destroy tissue and blood supply. Together, they produce one of the highest-mortality surgical emergencies in medicine.
The uses of HBOT in NSTI parallel those in gas gangrene: direct bactericidal action against the anaerobic component, toxin suppression, leukocyte potentiation, and antibiotic potentiation in previously hypoxic tissue. documented the complementary mechanism between restored leukocyte function and antibiotic therapy — applicable to all three infection indications.
| NSTI Evidence | Mortality with surgery alone: 30–50% for severe NSTI. With surgery plus HBOT: multiple series documenting reduction to 10–15%. A systematic review of NSTI outcomes documented that HBOT reduces mortality and amputation rates across the full spectrum of necrotising infection presentations. Fournier’s gangrene, which carries particularly high mortality without combined treatment, shows the most pronounced benefit. |
For the complete NSTI evidence and protocol, see our primary article on HBOT for necrotising fasciitis.
[IMAGE: uses-of-hyperbaric-oxygen-therapy-three-infection-mechanism-comparison.jpg | Alt: uses of hyperbaric oxygen therapy — three infection mechanism comparison anaerobic bactericidal leukocyte]
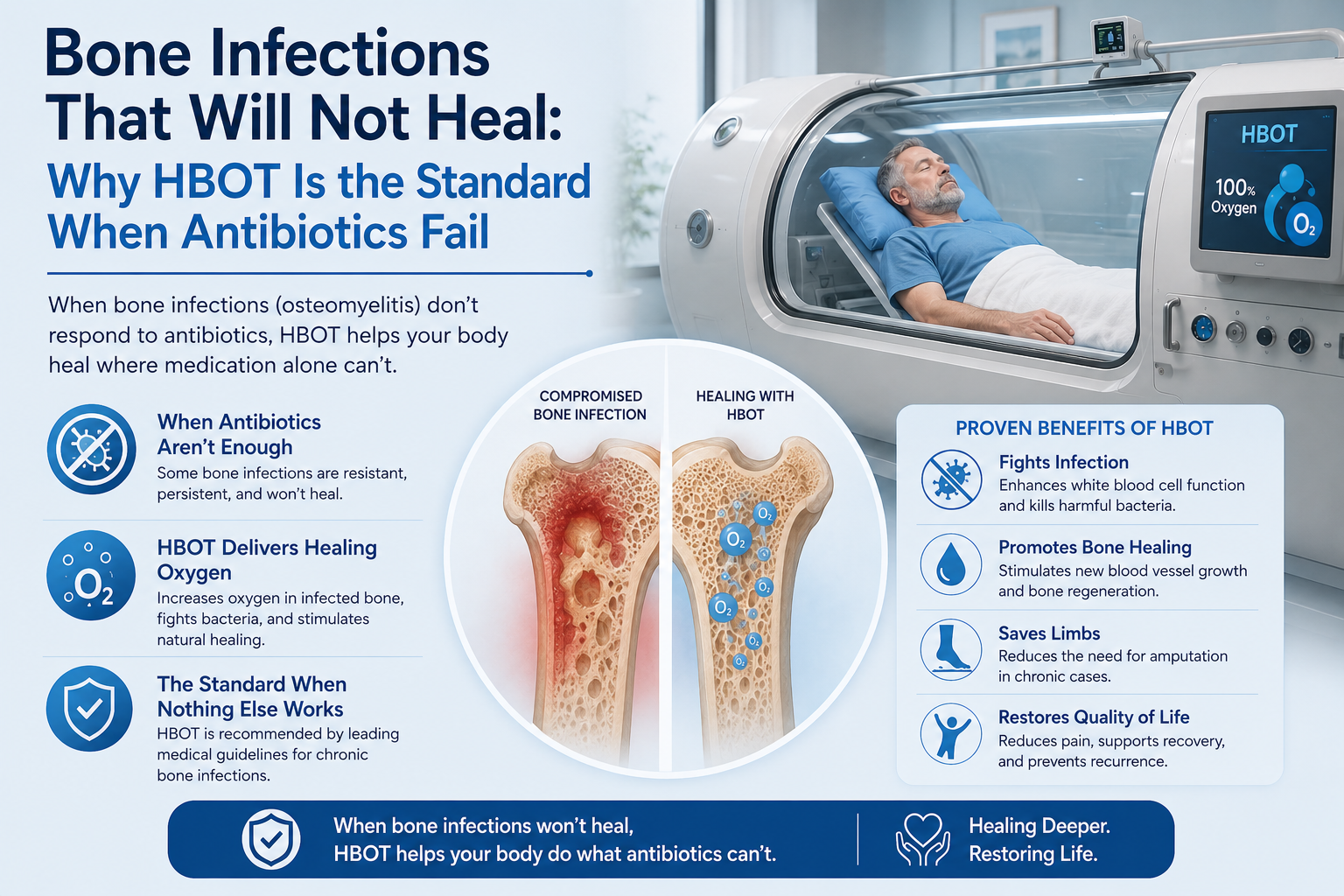
Infection Use 3 — Uses of Hyperbaric Oxygen Therapy in Refractory Osteomyelitis
Refractory osteomyelitis — chronic bone infection that has not responded to surgery and antibiotics alone — presents a different biological challenge from the acute infections above. The bacteria are typically Staphylococcus aureus, often in biofilm form. The bone tissue is hypovascular and hypoxic from years of chronic infection and repeated surgical intervention.
Three mechanisms make HBOT specifically relevant to refractory osteomyelitis, distinct from the acute infection uses:
- Antibiotic potentiation: aminoglycoside antibiotics require tissue oxygen tension above 30 mmHg for full efficacy — precisely the threshold that HBOT restores in chronically hypoxic bone
- Biofilm disruption: HBOT-generated reactive oxygen species disrupt bacterial biofilms that make Staphylococcal bone infections resistant to antibiotics alone
- Angiogenesis: the hyperoxic-hypoxic cycle triggers new blood vessel growth in hypovascular bone — rebuilding the delivery system for both immune cells and antibiotics
For the complete osteomyelitis evidence, see our article on HBOT for refractory osteomyelitis.
| Infection | Pathogens | Why HBOT Works | Protocol Pressure | Evidence Level |
| Gas Gangrene | Clostridium perfringens (obligate anaerobe) | Bactericidal at >200 mmHg; 50% toxin suppression; leukocyte potentiation | 2.5–3.0 ATA x 3 sessions/day 1; then BID | Strong — multi-decade series; 20% → <10% mortality |
| Necrotising Fasciitis | Mixed aerobic/anaerobic synergy | Kills anaerobic component; suppresses synergistic toxin production; immune restoration | 2.5 ATA BID x 10–20 sessions | Strong — systematic review; 30–50% → 10–15% mortality |
| Refractory Osteomyelitis | S. aureus, often biofilm | Antibiotic potentiation; biofilm disruption; angiogenesis in hypovascular bone | 2.0–2.4 ATA OD x 20–40 sessions | Strong — randomised controlled data; 50% reduction in surgical failure rate |
Three infections. Three different pathogen profiles. One shared vulnerability: they all depend on hypoxic tissue to resist treatment. The uses of hyperbaric oxygen therapy in infection are built entirely on destroying that dependency.
The Shared Mechanism Across All Three Infection Uses
Despite different pathogens and clinical presentations, all three infection uses of HBOT share a four-layer mechanism that differentiates them from conditions where standard treatment alone is sufficient.
Layer 1 — Direct Bactericidal or Bacteriostatic Effect
Obligate anaerobes die at high oxygen concentrations. Facultative anaerobes — including Staphylococcus in biofilm — have significantly reduced virulence in high-oxygen environments. HBOT at therapeutic pressure creates tissue oxygen concentrations lethal or growth-inhibiting to the organisms responsible for all three infections.
Layer 2 — Toxin Suppression
Clostridial alpha-toxin, streptococcal pyrogenic exotoxins in NSTI, and Staphylococcal toxins all have reduced production in aerobic environments. documented the measurable alpha-toxin suppression at therapeutic oxygen concentrations — confirming that the toxin reduction mechanism operates independently of direct bactericidal killing.
Layer 3 — Leukocyte Potentiation
Leukocyte killing requires tissue oxygen tension above 30 mmHg. Infected tissue in all three conditions has oxygen tension near zero. HBOT restores the oxygen environment that enables immune cells to function — converting a failing immune response into an effective one.
Layer 4 — Antibiotic Potentiation
Aminoglycosides — first-line for many serious infections — require aerobic conditions for transport across bacterial cell membranes. In hypoxic tissue, they are present but functionally inert. confirmed that HBOT restores aminoglycoside efficacy in hypoxic infected tissue — making the antibiotics that are already being administered finally work at their intended bactericidal level.
Infection Uses of HBOT in India — The Clinical Gap
India’s infectious disease burden makes all three HBOT infection uses clinically urgent. Agricultural injuries, road traffic accidents, tropical climate, high rates of diabetes, and varying wound hygiene create a high-incidence environment for all three conditions.
Yet awareness of HBOT as an adjunctive treatment in the infectious disease and emergency surgical community in India remains limited. Most Indian hospitals that perform surgery for gas gangrene, necrotising fasciitis, or osteomyelitis do not have on-site hyperbaric facilities — and many do not have established referral pathways to those that do.
The mortality gap between what is achievable with combined treatment and what is currently being achieved in Indian hospitals is measurable and preventable.
For HBOT facility access in India, see our guides to HBOT in Delhi and HBOT in Bangalore. For a complete national facilities guide, visit our HBOT near me India guide.
For guidance on whether HBOT for infections is covered by insurance in India, see our HBOT insurance India guide.
Frequently Asked Questions
Are all three infection uses formally recognised?
Yes. Gas gangrene, necrotising soft tissue infections, and refractory osteomyelitis are all formally recognised indications by both the US FDA and the Undersea and Hyperbaric Medical Society. They are covered in the UHMS Hyperbaric Oxygen Therapy Indications reference document and have been recognised for decades. They are not experimental or off-label uses — they are among the most established indications in the field.
Can HBOT replace surgery for these infections?
No for all three. Surgical debridement and drainage is non-negotiable for gas gangrene and necrotising fasciitis — removing the bacterial mass and necrotic tissue is essential and cannot be replaced by any non-surgical intervention. For osteomyelitis, surgical sequestrectomy where indicated is equally essential. HBOT addresses the biological environment that surgery cannot modify — not the infected tissue itself.
How does HBOT help when antibiotics have already failed?
Antibiotic failure in osteomyelitis and some NSTI cases is frequently not true resistance — it is environmental failure. The antibiotic concentration at the infection site is adequate. The tissue oxygen tension is not adequate for the antibiotic to work. HBOT restores the oxygen environment that allows the antibiotic to achieve its intended effect. This is why HBOT and continued antibiotic therapy together produce outcomes that neither achieves alone.
Is HBOT safe alongside broad-spectrum antibiotics?
Yes — there are no significant interactions between HBOT and standard broad-spectrum antibiotics used for serious infections. The combination is specifically additive: HBOT improves antibiotic efficacy in hypoxic tissue rather than interfering with antibiotic mechanism. The one antibiotic class requiring caution is bleomycin, used in some oncology protocols — this is a relative contraindication for HBOT, not relevant to infection management.
What are the side effects of HBOT in critically ill infection patients?
Ear barotrauma and sinus discomfort are the most common side effects. Oxygen toxicity seizures are rare at therapeutic pressures — fewer than 1 per 10,000 sessions. Critically ill patients are monitored continuously inside the chamber. For a complete side effects guide, see our article on hyperbaric oxygen therapy side effects.
Three Infections. One Oxygen Environment. The Same Treatment Logic.
Gas gangrene kills in hours. Necrotising fasciitis kills in days. Refractory osteomyelitis destroys limbs over months. All three do it through the same biological mechanism: creating and defending hypoxic tissue that resists every treatment directed at it.
These infection uses are the direct response to that mechanism. Not a supplement to standard care. Not a last resort. A specific, evidence-backed intervention that addresses the oxygen environment — the layer of the problem that surgery and antibiotics structurally cannot reach.
For patients, families, and clinicians navigating any of these three diagnoses in India — the question is not whether HBOT works. The evidence is established. The question is whether the referral pathway to a chamber can be activated in time.
The combined mortality data across all three infection uses is the most compelling case for HBOT in infection medicine: gas gangrene 20–30% → 5–10%, NSTI 30–50% → 10–15%, osteomyelitis surgical failure rate halved. These are not marginal improvements. They represent the difference that restoring oxygen to infected tissue actually makes.
For the full gas gangrene evidence, see our primary article on HBOT for gas gangrene. For the full 14-indication overview, explore our HBOT uses guide.
Three infections. One mechanism. Oxygen deprivation is the vulnerability. HBOT is the answer.