HBOT Skin Therapy for Grafts and Flaps: How Oxygen Rebuilds What Surgery Cannot
A skin graft is a second chance for the tissue that was lost. Whether that graft survives — and how well it integrates — depends on three conditions. HBOT skin therapy addresses all three simultaneously.
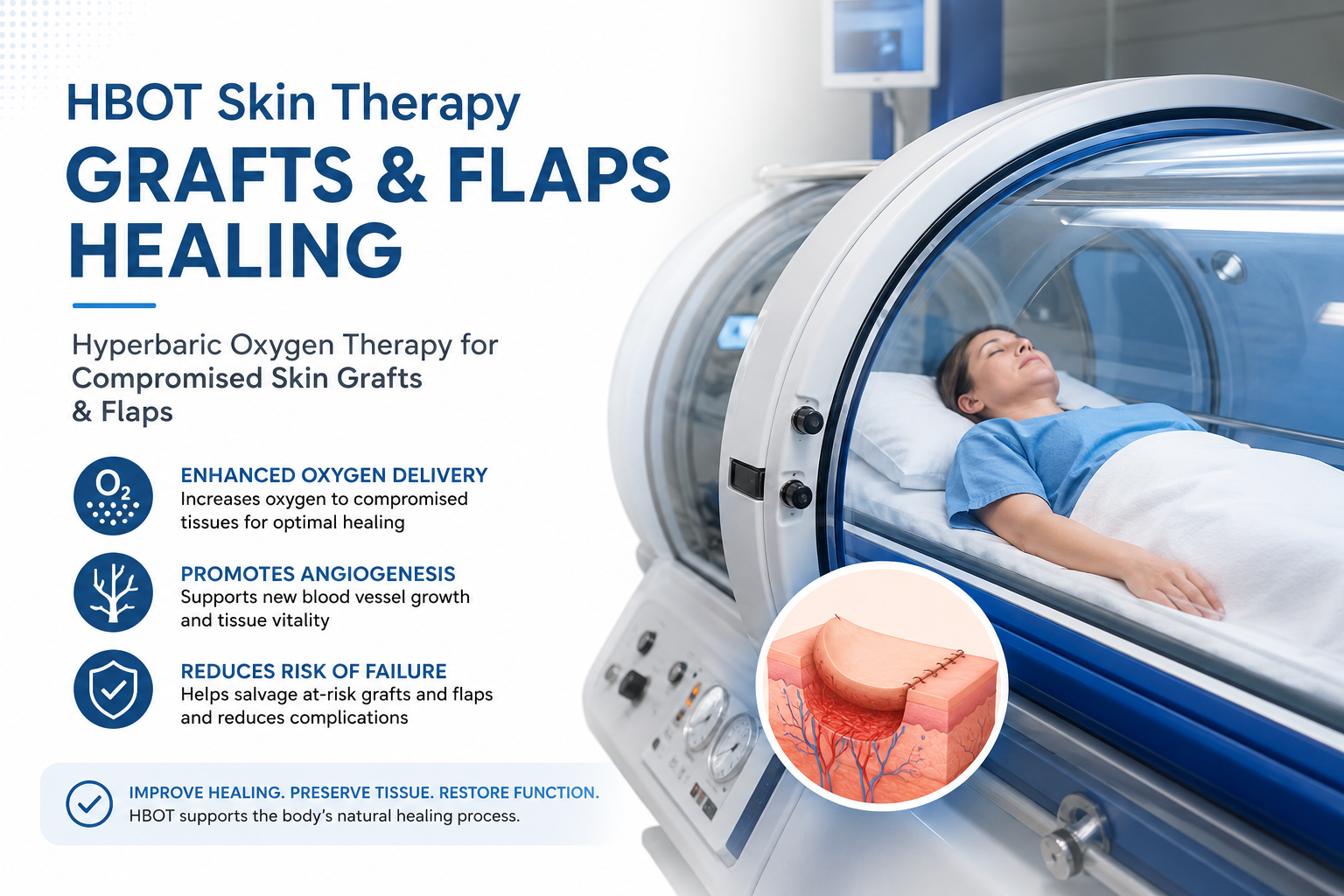
hbot skin therapy grafts flaps — hyperbaric oxygen therapy compromised skin graft wound healing
HBOT skin therapy changes the biological environment that determines whether a skin graft survives.
That is a precise statement — not a general claim. Skin graft failure has three identifiable biological causes: hypoxia in the graft bed, inadequate vascular ingrowth from bed to graft, and infection. These are not random events. They are predictable consequences of the clinical conditions that required the graft in the first place — compromised circulation, radiation damage, diabetes, or trauma.
Standard surgical technique cannot control these conditions after the graft is placed. The surgeon can optimise the graft bed, immobilise the graft, and apply appropriate wound care. But the oxygen level of the graft bed, the rate of new vessel formation, and the infection resistance of hypoxic tissue — these are biological processes that surgical technique alone cannot directly modify.
HBOT skin therapy modifies all three. Through plasma-dissolved oxygen, angiogenesis stimulation, and anti-infective tissue oxygenation — it converts a failing graft environment into a healing one.
The foundational mechanism behind how HBOT delivers oxygen to hypoxic tissue is explained at How HBOT Works. This article focuses specifically on the graft and flap evidence.
Compromised skin graft / flap: A skin graft or rotational/free flap that is at risk of partial or complete failure due to inadequate oxygen supply, poor vascular ingrowth, or infection at the graft bed. HBOT skin therapy is formally recognised by the UHMS and FDA as an indication for this condition — initiated within 72 hours of graft compromise for maximum benefit.
Why Skin Grafts and Flaps Fail — The Three Biological Causes
Skin graft take — the proportion of graft tissue that successfully integrates with the recipient bed — is determined by three biological variables operating simultaneously at the graft-bed interface.
| Failure Cause | Biological Mechanism | Clinical Context | HBOT Skin Therapy Role |
| Graft bed hypoxia | Insufficient O₂ for angiogenesis, fibroblast activity, and immune function | Diabetes, PVD, radiation, extensive tissue loss | Plasma-dissolved O₂ oxygenates bed independently of circulation |
| Inadequate angiogenesis | New vessels fail to grow into graft before it desiccates | Any graft — especially delayed take or large surface area | Hyperoxic-hypoxic cycle stimulates VEGF and new vessel growth |
| Graft bed infection | Bacteria destroy graft-bed interface before integration | Contaminated wounds, diabetic tissue, post-radiation beds | Bactericidal tissue O₂ + leukocyte potentiation reduces infection |
Skin graft failure is not surgical failure. It is biological failure — in three specific, addressable processes. HBOT skin therapy addresses each of them directly, simultaneously, in the same 90-minute session.
High-Risk Graft Patients — Who Fails Most Often
Graft failure is not evenly distributed. Certain patient groups carry dramatically higher risk — and these are precisely the patients for whom HBOT skin therapy evidence is strongest.
- Diabetic patients — peripheral vascular disease and impaired immune function create the worst possible graft bed environment
- Post-radiation tissue — radiation destroys the microvasculature permanently; graft beds in radiated tissue are hypovascular and hypoxic by default
- Elderly patients — reduced wound healing capacity, slower angiogenesis, and often concurrent vascular disease
- Large surface area grafts — the larger the graft, the longer the window before angiogenesis can sustain it; failure at the periphery is common
- Reconstructive flaps with arterial insufficiency — free flaps and pedicled flaps where the vascular pedicle provides marginal perfusion
How HBOT Skin Therapy Works — Three Simultaneous Mechanisms
Mechanism 1 — Plasma Oxygenation of the Hypoxic Graft Bed
At 2.0 to 2.4 ATA, oxygen dissolves into blood plasma at concentrations 15 to 20 times higher than at sea level. This plasma-dissolved oxygen reaches the graft bed through diffusion — independent of the compromised microvascular circulation that is causing the hypoxia.
The graft bed receives the oxygen it needs for angiogenesis, fibroblast collagen production, and immune cell function. The cells in the bed are not incapable — they are oxygen-starved. HBOT skin therapy delivers the input that makes them functional.
confirmed that HBOT delivers measurable wound healing improvement by restoring the oxygen gradient that compromised tissue cannot generate on its own — the same mechanism that makes HBOT skin therapy specifically effective in hypoxic graft beds.
Mechanism 2 — Angiogenesis: Building the Vessel Bridge from Bed to Graft
For a graft to survive beyond the first few days, new blood vessels must grow from the recipient bed into the graft tissue — a process called inosculation, followed by angiogenesis. This is the primary determinant of long-term graft take.
HBOT’s hyperoxic-hypoxic cycle — high oxygen during sessions, relative hypoxia between sessions — is the precise signalling environment that triggers vascular endothelial growth factor (VEGF) and vasculogenic stem cell mobilisation. documented that HBOT stimulates measurable neovascularisation in hypovascular tissue — creating the vessel bridge that graft survival depends on.
In post-radiation beds — where the microvasculature is permanently damaged and angiogenesis cannot occur spontaneously — HBOT skin therapy provides the only documented stimulus for new vessel formation. This is why it is specifically recommended for radiation injury patients undergoing reconstructive surgery.
Mechanism 3 — Anti-infective Protection of the Graft Interface
Graft bed infection destroys the delicate fibrin layer that anchors the graft before vascular ingrowth is established. In hypoxic tissue, immune function is impaired — leukocyte killing requires tissue oxygen tension above 30 mmHg to achieve its full bactericidal effect.
HBOT skin therapy restores tissue oxygen to levels that enable full immune function at the graft bed. The same tissue that was providing a hospitable environment for bacteria becomes hostile to them. For high-risk patients — diabetics, post-radiation, patients with contaminated wounds — this anti-infective mechanism is clinically essential, not supplementary.
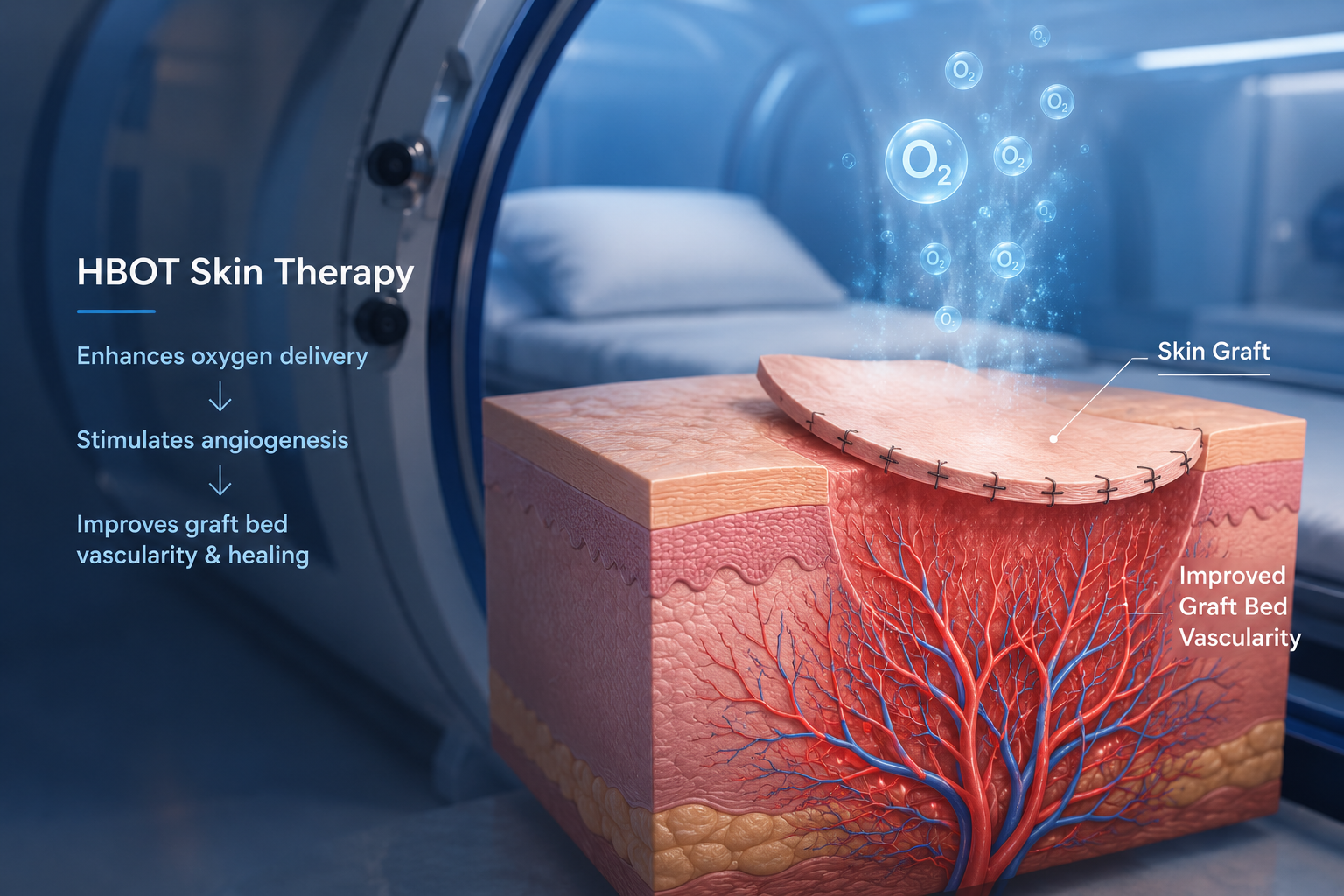
hbot skin therapy — angiogenesis graft bed mechanism hyperbaric oxygen wound healing
The 72-Hour Window — When HBOT Skin Therapy Must Begin
Timing is the critical variable in HBOT skin therapy for compromised grafts. The biology that determines graft survival is most active — and most reversible — in the first 72 hours after graft compromise is recognised.
| Time Since Graft Placement / Compromise | Graft Status | HBOT Skin Therapy Timing | Expected Benefit |
| 0–48 hours — pre-emptive in high-risk patients | Graft in place, showing early signs of poor take | Immediate initiation — prevent failure before it establishes | Highest — converts at-risk to successful graft |
| 48–72 hours — early compromise | Partial discolouration, marginal perfusion, early necrosis at edges | Urgent initiation — still within reversal window | High — can convert marginal areas; limit necrosis extent |
| 72–120 hours — established partial failure | Defined zones of failure, healthy central area | Still beneficial — limits extension; supports remaining viable graft | Moderate — cannot reverse established necrosis; limits spread |
| Beyond 5 days — established failure | Extensive necrosis, re-grafting planned | Supportive — prepares bed for second graft; reduces second failure risk | Lower for current graft; high value for next procedure preparation |
| UHMS & FDA Recognition | Compromised skin grafts and flaps are formally recognised HBOT indications by both the UHMS and US FDA. The clinical standard: initiate HBOT within 72 hours of identifying graft compromise. Sessions twice daily in the acute phase — daily thereafter until graft viability is established. HBOT is adjunctive to surgical wound care, not a replacement for re-grafting where required. |
The Evidence on HBOT Skin Therapy for Graft Survival
The evidence base for HBOT in compromised skin grafts and flaps is built across controlled studies, clinical series, and systematic reviews — with consistent findings across decades of hyperbaric plastic surgery literature.
A comprehensive 2024 review in documented that HBOT skin therapy significantly improves graft survival by simultaneously mitigating ischaemia and infection at the graft bed. Comparative data from the review showed reduction in major complications and improved healing outcomes in complex graft cases — specifically in the high-risk patient populations (diabetic, post-radiation, elderly) where standard care alone produces the highest failure rates.
The evidence on angiogenesis in graft beds specifically confirms that documented vasculogenic stem cell mobilisation and new vessel formation — the mechanism that converts a failing graft environment to a viable one. These cells arrive at the graft bed during the HBOT course and establish the vascular bridge that inosculation requires.
| 2024 Review Findings | MDPI 2024 comprehensive review — HBOT skin therapy in complex graft cases: significant improvement in graft survival in high-risk patients; simultaneous ischaemia and infection mitigation confirmed; reduction in major complications including need for re-grafting documented. The three-mechanism model — oxygenation, angiogenesis, anti-infective — is confirmed across the reviewed literature. |
HBOT Skin Therapy for Reconstructive Flaps — Pedicled and Free
HBOT skin therapy applies not only to split-thickness and full-thickness grafts but to the full range of reconstructive flap procedures used in plastic and reconstructive surgery.
Pedicled Flaps
Pedicled flaps maintain their blood supply through a vascular pedicle attached to the donor site. The distal portion of the flap — furthest from the pedicle — is most at risk of necrosis when perfusion is marginal. HBOT skin therapy oxygenates this distal zone through plasma diffusion and stimulates angiogenesis that extends the viable range of the flap.
Free Flaps
Free flaps are completely detached from their donor site and anastomosed to recipient vessels. The entire flap depends on the anastomosis for perfusion — making early post-operative vascular compromise particularly dangerous. HBOT skin therapy in the peri-operative period reduces the risk of ischaemia-driven flap failure and addresses reperfusion injury at the anastomotic site.
The reperfusion injury mechanism — documented by — is directly relevant to free flaps: when blood flow is restored through the anastomosis, the ischaemic flap tissue is at risk of a second wave of damage from reactive oxygen species and inflammatory cascades. HBOT modulates this response, protecting the flap tissue in the critical post-operative window.
For high-risk free flap patients — those with diabetes, radiation history, or peripheral vascular disease — HBOT skin therapy as a peri-operative adjunct is the evidence-based approach to improving flap survival rates.
HBOT Skin Therapy in India — Who Needs It and Where to Find It
The patient populations with the highest graft failure risk are disproportionately represented in India. Diabetes — with its peripheral vascular disease, neuropathy, and impaired immunity — affects approximately 77 million Indians and is the single largest risk factor for skin graft complications. Post-radiation tissue from India’s large cancer burden creates a significant population needing reconstructive surgery in compromised tissue. Burn grafting — given India’s high burn injury rate — represents another major volume.
Awareness of HBOT skin therapy among Indian plastic surgeons and reconstructive surgeons is growing but remains limited outside major academic centres. Most Indian hospitals performing reconstructive surgery do not have on-site hyperbaric facilities — meaning the 72-hour treatment window for compromised grafts cannot practically be met without advance referral planning.
For HBOT facility locations across India, see our guides to HBOT in Delhi and HBOT in Bangalore. For comprehensive facility guidance, see our HBOT near me India guide.
For the related evidence on HBOT skin therapy in diabetic wounds — which shares the ischaemic wound bed mechanism — see our detailed guide on HBOT for diabetic foot wounds. For radiation injury healing, see our article on HBOT for radiation injury.
Frequently Asked Questions
1. How quickly does HBOT skin therapy need to start for a compromised graft?
2. How many HBOT sessions are needed for a compromised graft?
3. Can HBOT skin therapy prevent graft failure from happening?
4. Does HBOT skin therapy work for both split-thickness and full-thickness grafts?
5. Is HBOT skin therapy covered by insurance in India?
6. Can HBOT skin therapy help with scars and wound healing beyond grafting?
The Graft Can Still Be Saved. The Window Is Now.
Skin graft failure is not inevitable for high-risk patients. It is predictable — and for the same reason that it is predictable, it is also preventable and reversible within the right window.
HBOT skin therapy addresses the three biological causes of graft failure simultaneously. Oxygenation. Angiogenesis. Infection resistance. Each mechanism operates independently. Together, they convert the failing graft environment into one where survival is possible.
The evidence is consistent. The UHMS and FDA both recognise it. For Indian patients undergoing skin grafting or reconstructive flap surgery in high-risk tissue — and for the surgeons planning those procedures — HBOT skin therapy is not an afterthought. It is part of the plan.
A graft placed on a well-oxygenated, vascularising, infection-resistant bed does not fail. HBOT skin therapy creates that bed. The biology is documented. The therapy is recognised. The window to use it is within 72 hours of compromise — and in high-risk patients, before compromise occurs.
To understand the full mechanism of how HBOT delivers oxygen to hypoxic tissue, visit How HBOT Works. For the complete 14-indication HBOT conditions evidence base, see our HBOT conditions reference.
The surgery gave the tissue a second chance. HBOT skin therapy makes that chance count.




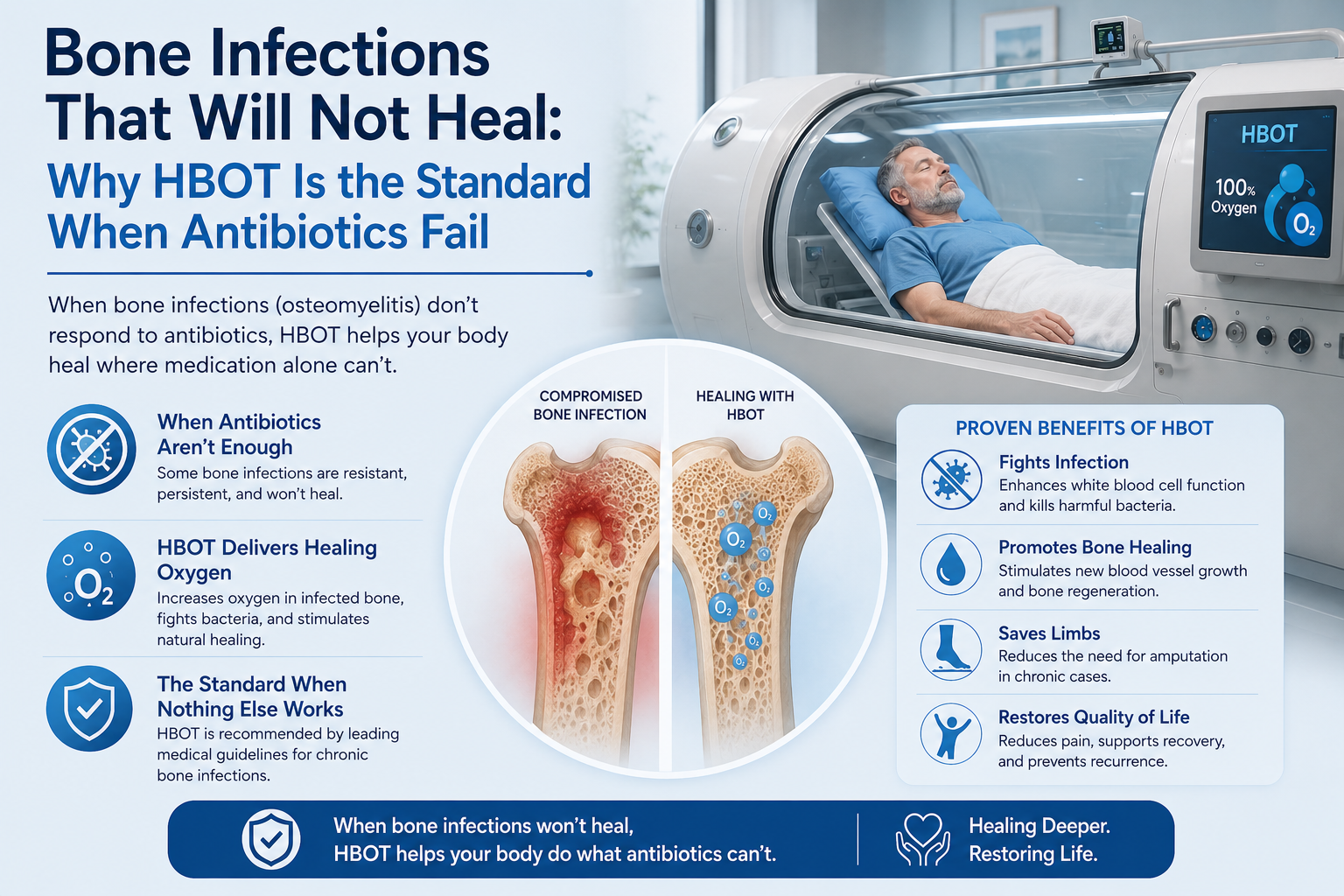

[…] The wound healing mechanism is shared with HBOT’s use in compromised skin grafts — covered in our article on HBOT skin therapy for grafts and flaps. […]