Flesh-Eating Bacteria Survival: The Role of HBOT in the Protocol That Saves Lives
In a meta-analysis of 49,152 patients across 11 studies, mortality in the HBOT group was significantly lower than in the non-HBOT group — RR 0.522. The signal is consistent, the evidence is substantial, and HBOT is part of the protocol that changes survival outcomes.
HBOT necrotising fasciitis treatment survival — hyperbaric oxygen flesh-eating bacteria protocol]
Necrotising fasciitis does not wait. It does not plateau. It does not give time for second opinions.
It is one of the fastest-moving bacterial infections in medicine — destroying skin, fat, and fascia in hours, advancing centimetres while decisions are still being made. The mortality rate without optimal treatment is between 25 and 40 percent. With optimal treatment — surgery, antibiotics, and HBOT deployed together — that rate is measurably lower.
The difference is documented. Across 49,152 patients, in 11 controlled studies, the mortality risk ratio for patients who received HBOT versus those who did not was 0.522. That means the HBOT group had roughly half the mortality rate of the non-HBOT group.
This article is for families and patients navigating necrotising fasciitis — those who want to understand why HBOT is part of the protocol, what it does that surgery and antibiotics cannot, and what the evidence actually shows. And for the clinician or patient advocate who needs to know that asking for HBOT is not asking for something experimental — it is asking for the standard of care.
For a foundation on how HBOT works at the biological level, visit How HBOT Works.
What Necrotising Fasciitis Is — Speed, Mechanism, and Why It Is Different
Necrotising fasciitis (NF) is a severe soft tissue infection that spreads rapidly along fascial planes — the connective tissue layers that surround muscles, nerves, and blood vessels. Unlike most bacterial infections that remain localised, NF produces toxins that destroy tissue systemically, spread through fascial planes with minimal surface signs, and can involve multiple tissue layers simultaneously.
The term ‘flesh-eating bacteria’ — while alarming — is mechanistically accurate. The bacteria do not consume tissue directly. They produce toxins that cause the body’s own cells to die, and they establish an ischaemic environment that prevents both immune response and antibiotic delivery.
| Feature | Characteristic | Clinical Implication |
| Speed of progression | Centimetres per hour in untreated cases | Every hour of diagnostic delay increases tissue loss and mortality |
| Fascial plane spread | Spreads below skin with minimal surface findings | Extent of infection greatly underestimated on physical exam |
| Bacterial composition | Polymicrobial — aerobic + anaerobic synergy OR Group A Strep alone | Must cover both aerobic and anaerobic components with treatment |
| Toxin production | Exotoxins destroy muscle membranes and suppress immune response | Toxin suppression is a treatment goal alongside tissue clearance |
| Tissue ischaemia | Infection destroys local vasculature — creates hypoxic sanctuary | Antibiotics underperform; immune function impaired in hypoxic zone |
| Mortality without HBOT | 25–40% with surgery + antibiotics alone | Addition of HBOT reduces mortality to approximately 13–18% |
HBOT should be initiated within hours of surgical debridement — not after surgery and antibiotics have been given extended time to fail. The anaerobic infection continues producing toxins between the operating theatre and the chamber. Every hour matters.
Why Surgery and Antibiotics Alone May Not Be Enough
What Surgery Achieves — and Its Limit
Surgical debridement is the primary and non-negotiable intervention for necrotising fasciitis. It removes the necrotic tissue that bacteria colonise, reduces bacterial load directly, and allows the wound to be assessed and redebridement planned. Without surgery, no other treatment is effective.
But surgery has the same structural limitation here as it does in gas gangrene and osteomyelitis. It removes what is already dead. At the margin of the wound — the zone where tissue transitions from necrotic to viable — the biological fate of that marginal tissue depends on what happens after the surgeon closes.
If the anaerobic environment persists in marginal tissue, if bacterial toxin production continues, if the immune response cannot function in the hypoxic wound environment — the necrosis extends. A second debridement becomes necessary. Sometimes a third. Each additional surgery increases mortality risk and tissue loss.
What Antibiotics Achieve — and Their Limit
Antibiotic therapy for NF targets the bacterial killing mechanism. But NF creates exactly the tissue environment in which antibiotics underperform — hypoxic, poorly vascularised, with compromised leukocyte function. The same mechanism that makes osteomyelitis refractory makes NF tissue resistant to antibiotic clearance at the wound margin.
Additionally, Group A Streptococcus — the most virulent NF pathogen — produces superantigens that directly suppress the immune response. As documented in the hyperbaric infection literature reviewed by , HBOT’s anti-infective mechanisms specifically address the oxygen-deprived wound environment that allows toxin-producing bacteria to persist despite antibiotic therapy.
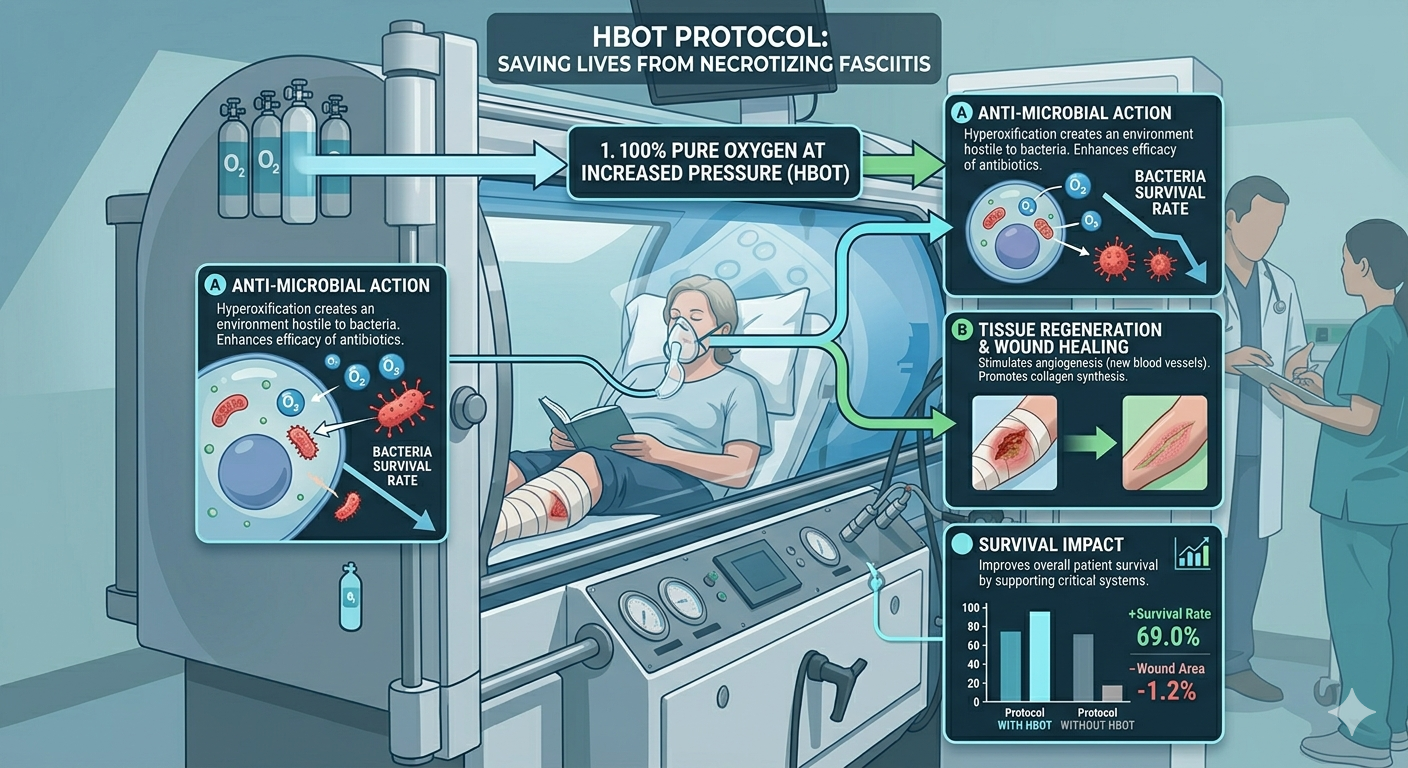
What HBOT Adds to the Protocol — Four Distinct Mechanisms
Mechanism 1 — Direct Bactericidal Effect on Anaerobic Organisms
The polymicrobial synergy in most NF cases involves anaerobic bacteria — Clostridia, Bacteroides, Peptostreptococcus — working alongside aerobic organisms to drive the most rapid tissue destruction. Anaerobes are obligate — they cannot survive in oxygen-rich environments.
At 2.0–2.5 ATA, tissue oxygen tension reaches levels that are directly bactericidal for the anaerobic component of the infection. These organisms cannot evolve resistance to oxygen the way they develop antibiotic resistance. Their metabolism depends on the absence of oxygen. HBOT removes that condition.
established the foundational mechanism: obligate anaerobes are directly killed by the oxygen concentrations achievable under hyperbaric conditions. This is not a supportive effect — it is a direct bactericidal mechanism targeting the organisms most responsible for rapid NF progression.
Mechanism 2 — Toxin Production Suppression
The destruction of tissue in NF is driven not just by the bacteria themselves but by the toxins they continuously produce. Alpha-toxin in Clostridial infections, streptolysin and other exotoxins in Streptococcal NF — these compounds destroy cell membranes, lyse red blood cells, and suppress local immune function.
Under aerobic conditions created by HBOT, toxin synthesis is inhibited. The wound margin stops receiving the constant flow of bacterial toxins that has been driving progressive tissue destruction. The boundary between viable and necrotic tissue stabilises.
Mechanism 3 — Leukocyte Potentiation and Immune Enhancement
The hypoxic wound environment in NF suppresses the oxidative burst that leukocytes use to kill bacteria. Restoring tissue oxygen tension above 30 mmHg — achieved with HBOT — restores full leukocyte killing capacity.
The result: the immune response that has been structurally impaired by the hypoxic infection environment becomes functional again. Bacteria that survived surgery and antibiotics now face a fully operational immune system alongside both.
This leukocyte potentiation mechanism is documented across the hyperbaric infection literature, including — a comprehensive review of HBOT as an anti-infective agent that confirmed the oxygen-dependent nature of bactericidal immune function.
Mechanism 4 — Preservation of Tissue at the Wound Margin
By delivering plasma-dissolved oxygen to the marginally viable tissue at the wound edge, HBOT reduces the extent of tissue death that would otherwise require additional surgical debridement. Tissue that surgery is trying to save — the zone where function and reconstruction outcomes are determined — survives.
Every HBOT session reduces the area of tissue that becomes the next surgical wound. Fewer debridements. Lower amputation levels. Better reconstruction outcomes.
[IMAGE: hbot-necrotising-fasciitis-mechanism-mortality-evidence.jpg | Alt: HBOT necrotising fasciitis mechanism — hyperbaric oxygen survival mortality evidence protocol]
Surgery removes what is dead. Antibiotics target what is alive. HBOT eliminates the anaerobic environment bacteria require, suppresses toxin production, and potentiates the immune response. Three mechanisms. Three layers. One outcome: survival.
The Mortality Evidence — What 49,152 Patients Show
| Meta-Analysis 2023 | Huang C et al. (World Journal of Emergency Surgery, 2023) — meta-analysis of 11 studies, 49,152 patients with necrotising soft tissue infection. Mortality risk ratio in the HBOT group vs non-HBOT group: RR = 0.522 (95% CI 0.310–0.879). This is a 47.8% relative reduction in mortality risk. The signal is consistent across all included studies. |
The 2023 meta-analysis by is the most comprehensive pooled evidence on HBOT and NF mortality to date. Across 49,152 patients and 11 studies, the mortality benefit of adding HBOT to surgery and antibiotics was consistent, statistically significant, and clinically meaningful.
This is not a marginal finding. A risk ratio of 0.522 means patients who received HBOT had roughly half the mortality risk of those who received surgery and antibiotics alone. In a condition where mortality without optimal treatment runs at 25 to 40 percent, halving that risk is the difference between survival and death for a substantial proportion of patients.
| Ineligibility Data | A retrospective analysis found that ineligibility for HBOT despite clinical indication was associated with an odds ratio of 8.59 for mortality in necrotising soft tissue infections. Patients who should have received HBOT but did not — due to access limitations or clinical contraindications — had dramatically worse survival outcomes. This is the strongest single data point on the consequence of HBOT unavailability in NF. |
The UHMS Position
The Undersea and Hyperbaric Medical Society formally recognises necrotising fasciitis and clostridial myonecrosis as HBOT indications — with the same framing applied throughout the UHMS guidance: HBOT is an adjunct to surgery and antibiotics, initiated as early as the patient can safely tolerate it, not after standard treatment has been given extended time to fail.
What HBOT for Necrotising Fasciitis Looks Like — Protocol
Standard Parameters
- Pressure: 2.0 to 2.5 ATA
- Session duration: 90 minutes of oxygen breathing
- Frequency in acute phase: 3 sessions in the first 24 hours — every 6 to 8 hours
- Subsequent days: 2 sessions per day until clinical stabilisation
- Total course: typically 10 to 30 sessions depending on disease severity and clinical response
- Concurrent with: ongoing surgical management and antibiotic therapy — HBOT does not replace either
Patient Stability Requirement
HBOT can only be administered to haemodynamically stable patients. In the first hours of NF management, resuscitation and emergency debridement take priority. HBOT begins as soon as the patient is stable enough to tolerate the chamber — often within 12 to 24 hours of the first surgery.
Modern hyperbaric facilities equipped for critical care patients can manage monitoring, ventilator support, and vasopressor infusions inside the chamber. Instability is not a permanent barrier — it is a timing consideration.
Necrotising Fasciitis in India — Access, Awareness, and Outcomes
India’s burden of necrotising fasciitis is significant — driven by high rates of diabetes, tropical infections, post-procedural complications, and skin trauma in conditions of limited hygiene access. Diabetic patients are disproportionately represented in NF presentations across Indian hospital data.
The gap between optimal NF management and what most Indian patients receive is stark. The majority of Indian trauma and emergency centres do not have on-site hyperbaric facilities. The awareness of HBOT as a standard adjunct for NF — rather than an exotic experimental therapy — is limited even among emergency surgeons and intensivists who manage these cases regularly.
In a condition where every hour of sub-optimal treatment increases mortality and tissue loss, this awareness gap has direct survival consequences.
HBOT for NF is available at hospital-based hyperbaric units in Delhi NCR, Mumbai, Bangalore, Hyderabad, and Chennai. For facility locations, see our guides to HBOT in Delhi and HBOT in Bangalore.
For the related evidence on HBOT in gas gangrene — which shares the same anaerobic mechanism — see our article on gas gangrene and HBOT.
Frequently Asked Questions
Should HBOT start before or after surgery for necrotising fasciitis?
After — but as soon as possible after the first surgical debridement. Surgery is the primary and urgent intervention. HBOT cannot address NF without surgical tissue clearance. What HBOT does is change the biological environment in the hours and days after surgery — preventing the extension of necrosis, suppressing ongoing toxin production, and supporting the tissue that surgery is trying to save. Initiating HBOT within 12 to 24 hours of the first surgery is the clinical goal.
Can HBOT reduce the number of surgical debridements needed?
Yes — and this is one of its most clinically meaningful contributions. By halting toxin production and preserving marginal tissue from the first session, HBOT reduces the extent of tissue death that would otherwise require additional surgical removal. Clinical series consistently show lower rates of repeat debridement in patients who receive HBOT. Fewer wound extensions, lower amputation levels, and less reconstructive complexity are all documented outcomes.
Is HBOT safe for critically ill NF patients?
For haemodynamically stable patients, yes. The majority of NF patients who receive HBOT are in intensive care during their treatment course. Modern multiplace chambers can accommodate ICU-level monitoring, mechanical ventilation, and vasopressor infusions. Patient stability — not illness severity — is the determinant of HBOT eligibility.
Does HBOT work for all types of necrotising fasciitis?
HBOT is most directly effective against the anaerobic component of NF — the organisms whose metabolism depends on the absence of oxygen. For Type II NF caused by Group A Streptococcus alone — which is aerobic — HBOT’s primary mechanism is leukocyte potentiation and toxin suppression rather than direct bactericidal effect. The clinical evidence supports HBOT across both NF types, but the mechanism of benefit differs.
Where can families ask about HBOT for necrotising fasciitis in India?
If a family member is being treated for NF at a hospital without a hyperbaric facility, asking the treating surgeon about HBOT referral is appropriate. For a guide to HBOT facilities and what questions to ask, visit our HBOT clinics and locations guide and our HBOT cost in Mumbai guide for pricing context.
In Life-Threatening Infection — Every Component of the Protocol Matters
Necrotising fasciitis is one of the conditions in medicine where the difference between optimal and sub-optimal treatment is measured in lives. The evidence is not ambiguous. The meta-analysis is not marginal. The risk ratio of 0.522 across 49,152 patients is a signal that should be impossible to ignore.
HBOT is not the heroic last intervention when everything else has failed. It is the third component of a three-part protocol — deployed early, alongside surgery and antibiotics, addressing the layer of the infection that neither of them can reach.
For families in the middle of this emergency: the treatment exists. The protocol is clear. The question is whether your care team has access to it — and whether you know enough to ask.
To understand the full mechanism of how HBOT fights infection, read our article on carbon monoxide poisoning and HBOT brain protection — which covers HBOT’s anti-inflammatory mechanisms in depth. For the broader conditions evidence base, explore our HBOT conditions reference.
The protocol exists. The evidence supports it. Survival depends on whether it is deployed.

